 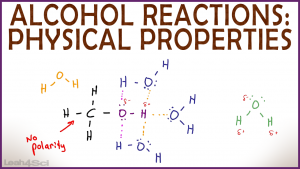
Of course, the first-row hydrides are left out of the discussion because of hydrogen bonding, which makes their boiling points anomalously high. (source: Greenwood & Earnshaw, Chemistry of the Elements 2nd ed., pp 557, 767) You can find an overview here on Chemistry LibreTexts. 5-10 ppm will cause discomfort.There are formulae you can use to calculate the strength of interactions between two molecules, which are derived from first principles. These are the ceiling valves that should not be exceeded during any part of the work exposure. The OSHA and ACGIH exposure limits for AHCl are 5 ppm or 7 mg/m3 and 7.5 mg/m3 TLV. the melting point of hydrogen is -259.87C and its boiling point is -252C. Therefore, the liquid hydrogen will need to be stored. Ingestion may cause severe acid burns of the mouth, throat, esophagus, and stomach with burning pain of the mouth, throat, chest, and abdomen. Hydrogen has a boiling point of 253C and the critical temperature for liquid hydrogen is 240C. Prolonged overexposure can cause dental erosion. The reaction, represented by the equation H2 + Cl2. For the red graph (Hydrogen halides), I understand that HF has the highest boiling point since it has hydrogen bonding, but why does HCl have a lower. 
Hydrogen chloride may be formed by the direct combination of chlorine (Cl2) gas and hydrogen (H2) gas the reaction is rapid at temperatures above 250 C (482 F). A solution of the gas in water is called hydrochloric acid. This large difference between the melting and boiling. High or prolonged inhalation exposures may cause delayed pulmonary edema with cough, chest discomfort, and difficulty in breathing. hydrogen chloride (HCl), a compound of the elements hydrogen and chlorine, a gas at room temperature and pressure. The melting point of water is 0 and its boiling point is 100 (at standard pressure or 0,987 atm). It is very irritating to the skin, eyes, and mucosal surfaces because of the rapid absorption by body moisture forming hydrochloric acid. Confined spaces should be well ventilated and equipment lines should be purged with inert gas or dry air until explosimeter tests show them to be free of hydrogen.ĪHCl is a gas at atmospheric pressure. Potentially explosive mixtures of air and hydrogen could be formed in confined spaces or closed equipment and lines, and in equipment and lines after opening. At -252.87C and 1.013 bar, liquid hydrogen has a density of close to 71 kg/m3. 
However, in the presence of water, AHCl attacks most metals with release of flammable hydrogen gas. Hydrogen turns into a liquid when it is cooled to a temperature below -252,87 C. More information on AHCl can be found in Pamphlet 99 and Pamphlet 160.Īnhydrous Hydrogen Chloride Release Fact SheetĪHCl will react with oxidizing agents to form chlorine gas. DO NOT ALLOW liquid AHCl to be trapped between closed valves - extremely high pressure can result. To exist as a liquid, H 2 must be cooled below its critical point of 33 K. Hydrogen is found naturally in the molecular H 2 form. In designing systems, care should be taken to eliminate, in so far as possible, places where liquid AHCl and Liquid/gas AHCl mixtures can become completely sealed between two closed valves because pressure could build up resulting in a gasket or line failure. Liquid hydrogen (H 2(l)) is the liquid state of the element hydrogen. Liquid AHCl has a high coefficient of thermal expansion. Because of these extreme values, the pressure in equipment containing liquefied AHCl must be closely monitored and controlled. 
The boiling point of AHCl at one atmosphere pressure (14.7 psia, 760 mmHg) is -85☌ (-121☏) and the vapor pressure of AHCl at ambient temperature of (20☌, 68☏) is 603 psia. It is very hydroscopic (attracts moisture) and in moist air, forms white fumes which are a mist of hydrochloric acid. It is readily absorbed in water to form hydrochloric acid. The boiling points of the hydrogen halides are as follows: Species Boiling point / C H C l 85.1 H B r 67.1 H I 35.1 (source: Greenwood & Earnshaw, Chemistry of the Elements 2nd ed., p 813) My textbook writes that this is due to an increase in the number of electrons, which increases the van der Waals forces between molecules. The melting and boiling points of para-hydrogen are about 0. Since the two forms differ in energy, the physical properties also differ. The ortho form cannot be prepared in the pure state. Hydrogen Chloride General Information about Hydrogen Chloride GasĪnhydrous hydrogen chloride, AHCl, is a colorless gas with a sharp, irritating odor. Normal hydrogen at room temperature contains 25 of the para form and 75 of the ortho form. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
